For years, Farrell Blum, struggled with sleep apnea. Like other adolescents with Down syndrome, his breathing would stop and start while he slept, the main symptom of the potentially dangerous condition, but he didn’t snore or seem sleepy during the day. While Blum had a continuous positive airway pressure (CPAP) machine, a common treatment for sleep apnea, he didn’t always like it.
Then his doctor mentioned a clinical trial for a device that works a bit like a tongue pacemaker. (Pacemakers typically are implanted into the heart to help control the heartbeat.) It would require surgery, but it meant Blum would no longer need to grapple with his CPAP machine's awkward and uncomfortable mask. His parents enrolled Blum in the study when he was 15 and were surprised by how the device transformed him.
“I saw a big improvement,” mom Janine Farrell-Blum, 58, from Long Island, told TODAY. “He was more alert in class. He was more cooperative. I can’t even begin to tell you — he was just a different person.”
Blum, now 21, was the second adolescent with Down syndrome to participate a phase one clinical study of the implantable device, a hypoglossal nerve stimulator, which is FDA approved for adults. It works by signaling the nerve that controls the tongue to move it forward, opening up the airway when the patient takes a breath.
While researchers were originally studying the hypoglossal nerve stimulator to make sure it was safe and effective in kids, they learned it seemed to provide other benefits, too. The paper was published in JAMA Otolaryngology — Head & Neck Surgery in April.
“Parents would come back to us and say, ‘Not only is this making their sleep better, but they’re doing better at school, and they’re speaking better, and they’re not as angry and aggressive and irritable,’” Dr. Christopher Hartnick, director of the pediatric otolaryngology division and Pediatric Airway, Voice and Swallowing Center at Massachusetts Eye and Ear, told TODAY.
Down syndrome and sleep apnea
As director of the Down Syndrome Program at Massachusetts General Hospital, Dr. Brian Skotko sees about 600 patients each year, who too often experience obstructive sleep apnea. The first step is often removing tonsils and adenoids, as enlarged tonsils and adenoids can cause sleep apnea in children. But sometimes in patients with Down syndrome, it doesn’t work.
People with Down syndrome develop sleep apnea for many of the same reasons as anyone else, but people with Down syndrome sometimes have smaller breathing pathways or larger tongues that can block their airways. Removing enlarged tonsils and adenoids helps many but not all.
“We have people with Down syndrome who are stuck with obstructive sleep apnea, and that could cause a lot of complications,” Skotko told TODAY. “(With) the treatment that we had … you have to wear a mask at night. And it’s hard enough for the neurotypical population to tolerate masking at night but, for people with Down syndrome — who have sensitivity issues, who have difficulty understanding it — it is just a real challenge.”
After a study about the hypoglossal nerve stimulator with the brand name Inspire was published, Skotko wondered if it could also work with his patients.
“It was working for the neurotypical (adult) population, and we said, ‘Oh, we have another population that really needs this therapy,’” he recalled. “Working with Dr. Hartnick, we were able to launch the study for people with Down syndrome.”
“Obstructive sleep apnea in any of us, if it goes untreated, can lead to brain fogging and some challenges in cognition,” Skotko added. “For people with Down syndrome that start with an intellectual disability, we do not want them to lose an extra 10 IQ points to obstructive sleep apnea.”
The clinical trial recruited 42 patients ages 10 to 22 to have the hypoglossal nerve stimulator implanted. Hartnick had to slightly modify the procedure so it worked better for younger patients.
In surgery, Hartnick inserts a processor under the skin above the right nipple so it can detect when the patient is trying to breathe. The processor sends a signal through a wire that runs under the skin near the jawline and is connected to the hypoglossal nerve, which can move the tongue forward and backward or sideways. It's important to make sure the device is connected to the right branch of the nerve to move the tongue forward, instead of side to side.
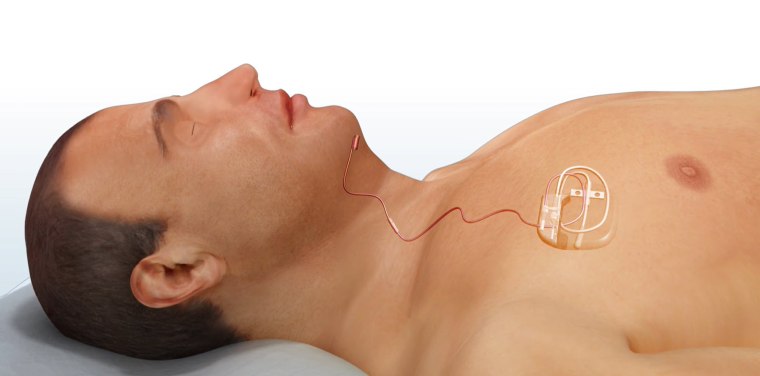
“As the child is drawing breath, the tongue is moving forward so the airway is unobstructed,” Hartnick explained.
The doctors continued to follow patients to make sure adjustments weren’t needed as they aged.
“As the children grew from not fully grown to fully grown, it did remain stable,” Hartnick said. “We felt comfortable with that.”
They also measured how much participants' sleep apnea improved using a metric called the apnea hypopnea index (AHI), Skotko said.
“Anything we could do to bring that number down is a step in the right direction," he explained. "Those who participated in the study that got a hypoglossal nerve stimulator implanted showed that their AHI was reduced, and for some of the people it was remarkably reduced.”
'Changed his life'
When Blum's mom, Janine Farrell-Blum, learned that the device would move Blum’s tongue when he slept, she wondered it would help with Blum's ability to communicate.
“That little thought bubble over my head was like, ‘Oh if we’re going to be stimulating his tongue for eight or nine hours a night, that’s just going to make his tongue muscles stronger, and his speech will improve,'” she said. “His fluency of speech is better.”
After about six months of using the device, Farrell-Blum noticed her son seemed like a different person.

“He’s sleeping better, and his whole body is better,” she said. “Our lives are so much richer because he feels better.”Blum, who takes boxing classes, swims, horseback rides and surfs, also realizes how important the device is for his health. Soon after his surgery, as he saw his mom packing for their upcoming trip, he made sure to include what he thought was an essential travel item — the remote control to the stimulator.
“That’s how important it was right away for him. He knew that it changed his life,” she said. “We’re in year seven. He uses it every night. I cannot think of a night that he has not used it.”
It's not yet clear when the device will be available to kids in the general public. Research is ongoing, and any parents interested interested in enrolling their child with Down syndrome in the clinical trial can find contact information for trial sites at ClinicalTrials.gov.
