The first children under 5 started getting COVID-19 vaccines on Monday in some locations, and the White House says parents of children as young as 6 months can schedule COVID-19 vaccines for their eligible kids across the country this week.
Shipments of the vaccine arrived in some locations over the weekend and some spots, including a Walgreens in South Carolina and another in New York City, offered appointments on Monday.
"These vaccines are exceedingly safe," White House COVID-19 Response Coordinator Dr. Ashish Jha told TODAY co-host Savannah Guthrie on Monday. Experience has shown that getting vaccinated "keeps kids out of the hospital," Jha said.
A Centers for Disease Control and Prevention (CDC) advisory committee endorsed both the Pfizer-BioNTech and Moderna’s COVID-19 vaccines for young children on Saturday. Following the endorsement, Director Dr. Rochelle Walensky signed off on COVID-19 vaccines, making the shots immediately available for eligible children ages 6 months to 5 years of age.
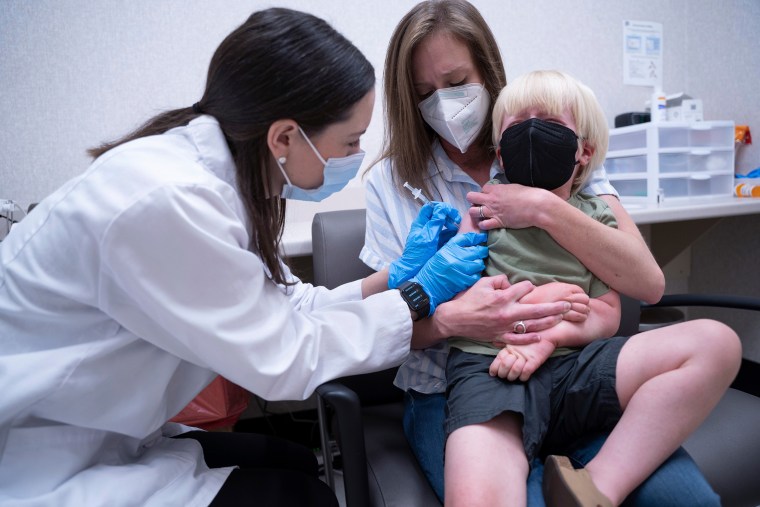
Three-year-old Fletcher Pack was one of the first little kids to get the vaccine in the U.S., the Associated Press reported. When his mom McKenzie Pack saw regulators moving toward approving the vaccine last week, she started looking for appointments. On Monday, Fletcher got vaccinated at a Walgreens in South Carolina.
“He’s never really played with another kid inside before,” McKenzie Pack said. “This will be a really big change for our family.”
She said that once her son is fully vaccinated, he can finally go bowling and visit the nearby children’s museum.
“It’s just relief,” said Pack. “With this vaccine, that’ll be his best shot at going back to normal and having a normal childhood.”
While the news will come as a relief to many parents with kids under 5, not everyone will be rushing to vaccinate their eligible children.
A Harris poll of 306 parents with children under 5 found that 73% of vaccinated parents said they're "likely to vaccinate their kids," while only 35% of unvaccinated parents said they would make a vaccination appointment for their child.
Related: COVID-19 vaccine timing, side effects and efficacy for kids under 5
A reported 61% of parents with children age 12-17 say that their child is vaccinated, according to a Kaiser Family Foundation survey. More than a quarter of parents say their child has had to quarantine because they tested positive or were exposed to someone who tested positive for COVID-19.
"We know that some number of kids do end up getting sick from COVID, unfortunately," Dr. Jha said. A reported 20,000 children have been hospitalized after contracting coronavirus, per CDC data.
"These vaccines are exceedingly safe," he added. "That's the biggest message: Now these vaccines have been given to millions and millions of kids — very, very safe. What I would say to parents is, talk to your family physician; talk to your pediatrician; talk to the people who take care of your kids and get their advice. That's probably the best way to move forward."

There have been over 2 million confirmed cases of COVID among children 6 months through 4 years of age, according to CDC data. Of those cases, over 200 children have died after contracting the virus.
COVID-19 is the fifth most common cause of death in children under 5.
"We do want to prevent infection, so it helps there," Dr. Jha said. "But where it's really useful — and we've seen this now with overwhelming data from kids 5-11 — is it keeps kids out of the hospital. A certain proportion of kids end up getting very sick, even kids without comorbidities, and the vaccines are terrific at keeping kids out of the hospital. So it helps some with infection but it helps a lot with prevention serious illness."
There are an estimated 18 million children under age 5 in the United States. The federal government has made 10 million vaccine doses available to states, tribes and other U.S. territories. Millions of additional doses are expected to be shipped in the next few weeks, according to the White House.
The Pfizer vaccine was authorized by the FDA for children age 6 months to 4 years, and is administered as a three-shot regimen. Moderna vaccine was authorized for children age 6 months to 5 years, and is given in two doses, four weeks apart.
Related:
